Maternal sepsis is “a life-threatening condition defined as organ dysfunction resulting from infection during pregnancy, child-birth, post-abortion, or the post-partum period” (WHO, 2016, manuscript in press). In Malawi post-partum sepsis accounts for 9.9% of all maternal deaths (Mataya, 2015).
In Malawi, a report on the Confidential Enquiry into Maternal Deaths highlighted deficiencies in the prompt recognition and inadequate management of maternal sepsis, concluding that the use of early warning scores and a structured approach to patient monitoring, alongside an educational programme to improve the recognition and management of maternal sepsis would likely improve sepsis care.
In high-income settings there is evidence that this approach can improve patient outcomes (Barochia et al., 2010; Dellinger et al., 2013; Damiani et al., 2015) however none of these interventions are specific to the maternal population or designed to be feasible in resource-poor settings (Bataar et al., 2010; Baelani et al., 2011). The development of a maternal sepsis bundle has been identified as an international “Priority Action” for the next five years (Jhpiego, 2015).
This study aims to determine whether it is feasible to introduce a complex intervention (including a bundled approach) to maternal sepsis care in resource-poor settings such as Malawi, to optimise an appropriate care bundle and to identify and carefully describe the facilitators and barriers to its implementation.
We have developed a treatment bundle entitled “FAST-M”. The ‘FAST-M’ acronym stands for consideration of the following treatment components:
- Fluids
- Antibiotics
- Source Control
- Transport
- Monitoring (of mother and newborn)
The complex intervention that we will implement consists of three components:
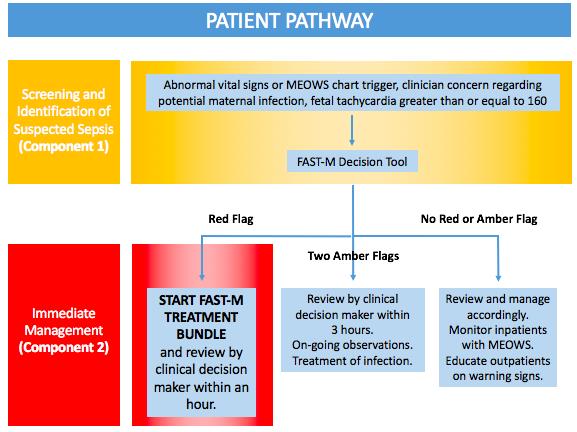
1) Modified Early Obstetric Warning Score and FAST-M decision tool
An early warning score is a tool used to track a patient’s physiological parameters over time with the aim of identifying early deteriorations in their health and intervening before they become very unwell. Whilst widely adopted in the UK, early warning scores are less widely used elsewhere although have been shown to be feasible to implement in resource-poor settings (Merriel et al., 2016).
In our study we will ask healthcare practitioners to routinely take and record patient observations (heart rate, respiratory rate, oxygen saturations (where available), blood pressure, conscious level, urine output and temperature) and fetal heart rate (if applicable). In the inpatient setting these observations will be charted on an early warning score chart. In the outpatient setting, these will be recorded on an outpatient sticker which can be inserted into the patient’s health passport. Abnormal observations (indicated by a single red or two amber thresholds) will trigger a review by a clinical decision maker.
The FAST-M decision tool is a tool that has been designed to act as an aide memoire to guide healthcare workers through the decision making process to enable prompt diagnosis of maternal sepsis. All patients with abnormal observations on the early warning score charts as well as those in whom there is concern about a potential infection and those with a fetal tachycardia will trigger the healthcare workers to start using the FAST-M decision tool. The tool will then guide the healthcare worker as to whether the patient has maternal sepsis.
2) FAST-M treatment bundle
Those patients that are highlighted as having maternal sepsis by the FAST-M decision tool will be prompted to initiate treatment for maternal sepsis using the FAST-M treatment tool. The treatment tool guides the healthcare practitioner through the components of the treatment bundle, again acting as an aide memoire to ensure timely treatment within an hour of recognition of maternal sepsis.
3) Staff training and implementation programme
Healthcare practitioners involved in the study will attend a training day focusing on the education around the use of the early warning score charts, FAST-M decision tools and the FAST-M treatment tool.
Patient pathway